Probe into arsenic content in cold tablets
 0 Comment(s)
0 Comment(s) Print
Print E-mail Shanghai Daily, March 29, 2013
E-mail Shanghai Daily, March 29, 2013
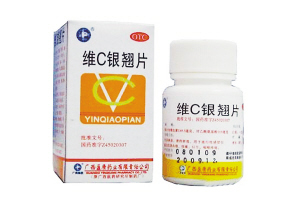
The Guangxi Zhuang Autonomous Region Food and Drug Administration is investigating a leading pharmaceutical company's local subsidiary after reports that its Yinqiao cold tablets mixed with vitamin C, which are available over-the-counter, may contain arsenic and mercury.
The Guangxi FDA has asked the Guangxi Yingkang Pharmacy Co to stop producing and marketing the product until further notice.
A China Central Television report on Tuesday claimed that Yingkang's tablets contain arsenic and mercury because sulphur is used to steam the raw materials.
The parent company, Guangzhou Pharmaceutical Holdings Limited, said that the allegedly chemical-laced tablets produced by its subsidiary have been taken off the shelves and sealed for further investigation.
The company's supplier of raw materials is also under the FDA's scanner.
A Guangxi Yingkang executive, surnamed Liang, said the company was cooperating with authorities in their investigation, Beijing Times reported yesterday.
One of the raw materials of the tablet, wild honeysuckle flower, is a popular traditional prescription used for coughs, sore throats and headaches.






Go to Forum >>0 Comment(s)